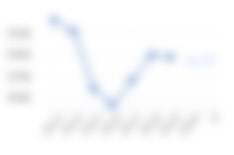
AVLN — Avalyn Pharma Share Price
Growth & Value
| 12m Forecast Rolling | Industry | Market | |
|---|---|---|---|
| PE Ratio (f) | n/a | ||
| PEG Ratio (f) | n/a | ||
| EPS Growth (f) | n/a | ||
| Dividend Yield (f) | n/a | ||
| Valuation (ttm) | Industry | Market | |
|---|---|---|---|
| Price to Book Value | n/a | ||
| Price to Tang. Book | n/a | ||
| Price to Free Cashflow | n/a | ||
| Price to Sales | n/a | ||
| EV to EBITDA | n/a | ||
Shareholder Activity
| Type | Buy / Hold / Sell |
|---|---|
| Institutions | |
| Directors | |
| Community |
Guru Screens
Quality
| Name | Industry | Market | |
|---|---|---|---|
| Return on Capital | n/a | ||
| Return on Equity | n/a | ||
| Operating Margin | n/a | ||
Financial Summary
| Year End null | Unit | null | null | null | null | null | nullE | nullE | CAGR / Avg |
|---|---|---|---|---|---|---|---|---|---|
| Total Revenue | $m | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
| Operating Profit | m | ||||||||
| Net Profit | m | ||||||||
| EPS Reported | |||||||||
| Diluted Normalised EPS | |||||||||
| EPS Growth | % | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
| PE Ratio | x | ||||||||
| PEG | |||||||||
| Profitability | |||||||||
| Operating Margin | % | ||||||||
| ROA | % | ||||||||
| ROCE | % | ||||||||
| ROE | % | ||||||||
| Cashflow | |||||||||
| Op. Cashflow ps | |||||||||
| Capex ps | |||||||||
| Free Cashflow ps | |||||||||
| Dividends | |||||||||
| Dividend ps | |||||||||
| Dividend Growth | % | ||||||||
| Dividend Yield | % | ||||||||
| Dividend Cover | x | ||||||||
| Balance Sheet | |||||||||
| Cash etc | m | ||||||||
| Working Capital | m | ||||||||
| NFA | m | ||||||||
| Net Debt | m | ||||||||
| Book Value | m | ||||||||
| Diluted Weighted Average Shares | m | ||||||||
| Book Value ps | |||||||||
Other Ratios
| Leverage (ttm) | Total | - Intang | + Pension |
|---|---|---|---|
| Gross Gearing | |||
| Net Gearing | |||
| Cash / Assets |
| Liquidity (ttm) | |
|---|---|
| Curr. Ratio | |
| Quick Ratio | |
| Interest Cov. | |
| Efficiency (ttm) | |
|---|---|
| Asset Turnover | |
| Recs Turnover | |
| Stock Turnover | |
Recent History
| Latest interim period vs. prior period | Industry | Market | |
|---|---|---|---|
| Sales Growth | |||
| EPS Growth | |||
| 3yr Compound Annual Growth Rate | Industry | Market | |
|---|---|---|---|
| Sales CAGR | |||
| EPS CAGR | |||
| DPS CAGR | |||
Profile Summary
Avalyn Pharma Inc. is a clinical-stage biopharmaceutical company engaged in inhaled therapies to transform the treatment paradigm of serious, rare respiratory diseases with significant unmet needs. The Company's pipeline is focused on treating pulmonary fibrosis. It has three approved oral antifibrotic therapies for pulmonary fibrosis: pirfenidone; nintedanib, and nerandomilast. Its clinically advanced candidates, AP01 and AP02, are inhaled formulations of pirfenidone and nintedanib for the treatment of progressive pulmonary fibrosis and idiopathic pulmonary fibrosis, respectively. It has completed ATLAS, a Phase 1b clinical trial of AP01 in patients with IPF. AP02 is being evaluated in AURA, a global Phase II clinical trial for the treatment of IPF. It is also advancing AP03 into a Phase I clinical trial. AP03 is a fixed-dose combination of inhaled pirfenidone, and nintedanib designed to deliver dual antifibrotic mechanisms with the potential for additive or synergistic benefit.
Directors
- Last Annual
- Last Interim
- Incorporated
- May 27th, 2011
- Public Since
- April 30th, 2026
- Sector
- Biotechnology & Medical Research
- Industry
- Healthcare
- Exchange
NASDAQ Global Select Market
- Address
- 105 W First St, BOSTON, 02127
- Web
- https://www.avalynpharma.com
- Phone
- +1 2067070340
- Auditors
Upcoming Events for AVLN
Similar to AVLN
4D Molecular Therapeutics
NASDAQ Global Select Market
Aardvark Therapeutics
NASDAQ Global Select Market
AbCellera Biologics
NASDAQ Global Select Market
AbSci
NASDAQ Global Select Market
Acadia Pharmaceuticals
NASDAQ Global Select Market
FAQ
As of Today at 01:27 UTC, shares in Avalyn Pharma are trading at $30.62. This share price information is delayed by 15 minutes.
Shares in Avalyn Pharma last closed at $30.62 and the price had moved by over the past 365 days. In terms of relative price strength the Avalyn Pharma share price has matched the S&P500 Index by over the past year.
There is no consensus recommendation for this security.
Find out moreAvalyn Pharma does not currently pay a dividend.
Avalyn Pharma does not currently pay a dividend.
Avalyn Pharma does not currently pay a dividend.
To buy shares in Avalyn Pharma you'll need a share-dealing account with an online or offline stock broker. Once you have opened your account and transferred funds into it, you'll be able to search and select shares to buy and sell. You can use Stockopedia’s share research software to help you find the the kinds of shares that suit your investment strategy and objectives.
As of the previous close price of $30.62, shares in Avalyn Pharma had a market capitalisation of .
Here are the trading details for Avalyn Pharma:
- Country of listing: United States
- Exchange: NSQ
- Ticker Symbol: AVLN
Based on an overall assessment of its quality, value and momentum Avalyn Pharma is currently classified as a . The classification is based on a composite score that examines a wide range of fundamental and technical measures. Stock are classified on the the following spectrum: Super Stocks, High Flyers, Contrarians, Turnarounds, Neutral, Value Traps, Momentum Traps, Falling Stars, and Sucker Stocks. For more information, learn about our StockRank Styles.
We could not find analyst target price data for this security.
Find out moreAn important predictor of whether a stock price will go up is its track record of momentum. Price trends tend to persist, so it's worth looking at them when it comes to a share like Avalyn Pharma. Over the past six months, its share price has matched the S&P500 Index by .
As of the last closing price of $30.62, shares in Avalyn Pharma were trading +5.68% higher than their 200 day moving average. You can read more about the power of momentum in assessing share price movements on Stockopedia.
The Avalyn Pharma PE ratio based on its reported earnings over the past 12 months is null. The shares last closed at $30.62.
The PE ratio (or price-to-earnings ratio) is the one of the most popular valuation measures used by stock market investors. It is calculated by dividing a company's price per share by its earnings per share.
The PE ratio can be seen as being expressed in years, in the sense that it shows the number of years of earnings which would be required to pay back the purchase price, ignoring inflation. So in general terms, the higher the PE, the more expensive the stock is.
We do not have data on Avalyn Pharma's directors